Findings
Figure 1: CT scan reveals unilateral hyperdensity of the left lentiform nucleus and the head of the left caudate nucleus with sparing of the intervening internal capsule that corresponds to abnormal gemistocytic astrocyte production. No significant abnormal enhancement is identified.
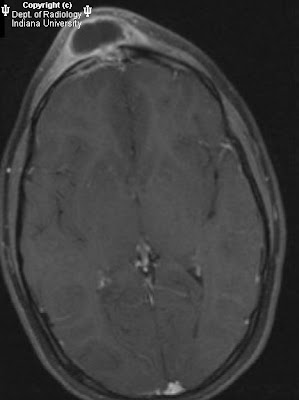
Figure 2: MR images demonstrate hyperintense T1 and hypointense T2 and diffusion weighted image signals in the same distribution as the CT abnormality. There is no significant abnormal enhancement and no mass effect.
Figure 3: Follow-up imaging performed 14 weeks after initial presentation demonstrates preserved high signal in the left basal ganglia without mass effect or enhancement. T2-weighted signal has changed from low to high intensity, perhaps representing interval gliosis.
Diagnosis: Non-ketotic, hyperglycemic, hemichorea
The differential diagnosis for the new-onset hemichorea includes: stroke, hemorrhage, tumor, infectious disease, neurodegenerative disorders and non-ketotic, hyperglycemic hemichorea.
Particular to the diagnosis of NHH, the characteristically unilateral transient extrapyramidal motions resolve following glycemic control.
Cerebral images of these patients consistently have shown unilateral CT hyperattenuation and unilateral MRI T1-weighted hyperintensity in the striatum contralateral to the side of the transient, extrapyramidal motion disorder.
Typically, an elderly diabetic patient presents with new-onset hemichorea as well as glucose levels ranging from 400 to 600 mg/dl with an HBA1c greater than 13%.
Non-ketotic, hyperglycemic hemichorea (NHH) was first described by Rector, et al. in 1982. Thirty-three cases of NHH have been identified in our recent review of literature. The most commonly reported location of a lesion has been in the putamen. Kumral, et al. have suggested that the anterior putamen needs to be involved for transient extrapyramidal motions to occur. The distinct imaging findings are hyperattenuation on CT and hyperintensity on T1-weighted MRI. This Case in Point’s images concur with these distinct image findings.
The onset of NHH has been attributed to petechial hemorrhage with blood-brain barrier breakdown, cerebral ischemia leading to dysfunction of the GABAnergic projection neurons and gliosis. Blood glucose levels of 159 to 647 mg/dl have initiated non-ketotic hyperglycemia resulting in hemichorea. More commonly reported values exceed 500 mg/dl.
There is accumulating consensus on the pathophysiology of NHH. Human autopsy, animal studies, human biopsy and MR spectroscopy evidence that NHH results in mild infarction with gliosis and concurrent accumulation of pathologically swollen, nucleus-eccentric astrocytes, known as gemistocytes. MRI findings have been attributed to gemistocyte deposition along axons. Rat model and human autopsy histopathology have revealed T1-weighted, hyperintense, gliotic brain tissue with abundant gemistocytes.
This Case in Point documents new hyperintensity in the areas of previous hypointensity on follow-up T2-weighted MRI and FLAIR images. It is plausible such images represent areas of delayed gliosis or scar tissue formation.