Findings
FindingsFigure 1: Contrast enhanced CT shows an enhancing soft tissue mass at the jugular foramen.
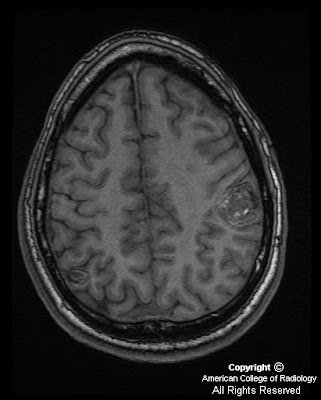
Figure 2 and Figure 3: Pre and post contrast axial T1 weighted MR images demonstrate enhancement of the mass at the jugular foramen with “salt and pepper” signal drop out from vessels (Figure 3).
Figure 4: Coronal T1 post contrast MR image again demonstrates an enhancing mass at the jugular foramen with “salt and pepper” signal drop out from vessels.
Differential Diagnosis of common jugular foramen lesions:
- Glomus jugulare
- Dehiscent jugular bulb / Enlarged Jugular bulb(normal variant)
- Meningioma
- Metastasis
- Schwannoma
Diagnosis: Glomus jugulareGlomus tumors are slow growing, encapsulated, hypervascular benign tumors which are locally invasive and originate from paraganglia cells which are neural crest progenitor cells of neuroectodermal origin. They are also referred to as paragangliomas, chromaffinomas or chemodectomas. Frequently located near nerves and vessels, their distribution can be widespread, including the periaortic area, trachea, larynx, mandible, nose, ciliary ganglion, and Fallopian canal.
Glomus tumors constitute less than 1% of all head and neck tumors. In the head and neck, they most commonly arise from within the carotid body (bifurcation), vagus nerve, middle ear, and jugular foramen. Specifically, glomus jugulare tumors originate from glomus bodies located within the wall (adventitia) of the jugular bulb. Additionally, they can be associated with either the auricular branch of the vagus nerve (Arnold nerve) or the tympanic branch of the glossopharyngeal nerve (Jacobson nerve). Arising from the jugular foramen of the temporal bone, these tumors grow within the temporal bone via pathways of least resistance, such as air cells, vascular lumens, skull base foramina, and the eustachian tube. Erosions occur frequently involving the jugular fossa and posteroinferior petrous bone. As the tumors grow they may also extend into local extracranial structures including the mastoids, occipital bone, sigmoid and petrosal sinuses. Intracranial extension with neural infiltration is also common.
Although most are sporadic, glomus tumors can be familial (autosomal dominant inheritance and incomplete penetrance). Multifocal lesions are found in 3-10% of sporadic cases and in 25-50% of familial cases. Malignant degeneration is most frequent with glomus vagale (16%) and least in jugulotympanic (4%) tumors. Metastases from glomus tumors occur in approximately 4% of cases. About 10% of head and neck paragangliomas are multicentric but bilaterality in glomus jugulare is only about 1-2%. Multiple glomus tumors can be associated with familial syndromes like multiple endocrine neoplasia (MEN) 1 and 2 , neurofibromatosis and von Hippel –Lindau disease.
Additionally, functional tumors may be found in 1-3 % of tumors producing clinically significant levels of catecholamines (norepinephrine or dopamine) with symptoms mimicking a pheochromocytoma. Rarely, an association with pheochromocytoma, parathyroid adenoma, and thyroid carcinoma may occur.
Due to their painless, slow growing nature, presentation usually manifests late in the course of disease with complaints ranging from lower cranial nerve palsies, Horner’s syndrome and hoarseness to other neuro-otologic symptoms including pulsatile tinnitus (most commonly associated symptom of glomus jugulare specifically). The glomus tumors are more frequently seen in patients with chronic hypoxia (COPD) and in those living at high altitudes. Due to the location and extent of disease which is often found at diagnosis, treatment and management is often very challenging. Patients with cervical glomus lesions typically have a more favorable prognosis. When identified in the temporal bone, however, there is often an increased rate of recurrence, residual tumor, and neurovascular compromise. Definitive treatment is surgical, however, radiation therapy may be employed for palliation for extensive tumors not amenable to surgery.
Imaging characterization of the location and extent of tumor involvement is critical in surgical planning and assessment of operative morbidity and mortality. Classic CT findings associated with these lesions include an enhancing soft-tissue mass in the carotid space, jugular foramen, or tympanic cavity. Cystic changes are not described in these tumors unlike in schwannomas from which glomus jugulare needs to be differentiated . Additionally, osseous erosion (a typical finding with the jugular type) adjacent to the tumor is best demonstrated on CT. By contrast the meningiomas, schwannomas and prominent jugular bulb tend to produce smooth expansion of bony outlines of the jugular fossa. MRI is superior to CT scan and typically demonstrates a "salt-and-pepper" appearance at standard spin-echo sequences due to signal drop out from extensive vascularity which manifests as an intense blush at angiography.