Findings
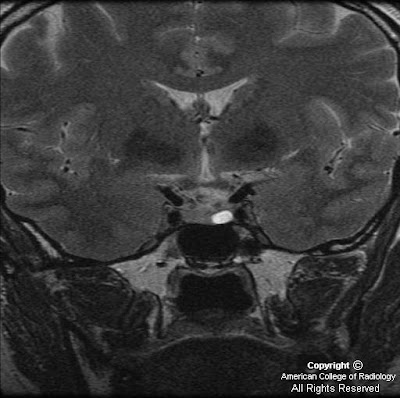
FindingsMRI of the thoracic and cervical spine show increased T2 signal within the posterior columns and the lateral corticospinal tracts on axial imaging. On sagital imaging, a longitudinal T2 signal abnormality is noted within the dorsal cord. There was no significant contrast enhancement status post gadolinium administration.
Differential diagnosis:
- Subacute combined demyelination
- Multiple sclerosis
- Astrocytoma
- Spinal cord infarction
- Infectious myelitis
Diagnosis: Subacute combined degeneration.Vitamin B12 deficiency is the result of a malabsorption syndrome and can affect the brain, optic nerves, peripheral nerves and spinal cord. When patients present with myelopathy, such as sensory disturbances, weakness, and spasticity, it is known as subacute combined degeneration (SCD).
Clinical presentation of SCD is caused by dorsal column, lateral corticospinal tract, and sometimes lateral spinothalamic tract dysfunction. Patients initially present with paresthesia in the hands and feet, which can progress to sensory loss, gait ataxia, and distal weakness particularly within the legs. If the disease goes untreated, ataxic paraplegia may evolve. On physical exam, there is a loss of vibratory and joint position sense, weakness, spasticity, hyperreflexia, and extensor plantar responses. The diagnosis of B12 deficiency is made by serologic studies showing low serum B12 level; if the B12 level is borderline, elevated levels of homocysteine and methylmalonic acid help cinch the diagnosis.
NeuroradiologyRadiologic manifestations of SCD may be seen on MRI imaging, primarily within the cervical and thoracic spine, and include the following spinal manifestations:
- Mild spinal cord expansion and hypointensity on T1 weighted imaging.
- Increased T2 signal intensity primarily within the dorsal columns +/- lateral columns.
- Longitudinal dorsal cord T2 signal abnormality.
- Inverted "V" or "rabbit ears" T2 signal intensity within the dorsal spinal cord on axial imaging.
- Possible mild dorsal column contrast enhancement, with enhancement signifying breakdown of the blood nerve barrier.
Differential considerationsAlthough these MRI findings are consistent with SCD, they are nonspecific and include a broad differential diagnosis: demyelinating disorders, infectious etiologies, inflammatory conditions, ischemia, contusion, and neoplasms. However, SCD can be distinguished from other differentials given its bilateral nature of T2 signal abnormality over multiple levels that is confined to specific white matter spinal tracts. There can be partial to full reversal of MRI abnormalities following B12 therapy, with fifty percent of patients fully recovering and with the greatest recovery occurring when treatment is began in the early stages of the disease.
Multiple sclerosis is a demyelinating disorder with multiple lesions separated in time and space. MRI spine findings included increased T2 signal intensity and hypo or isointense T1 signal within lesions. These lesions, however, are more focal and well-circumscribed in nature compared to the contiguous lesions of SCD and show homogenous, nodular, ring enhancement of acute and subacute lesions status post contrast administration. Multiple sclerosis lesions rarely spans more than one or two vertebral segments and are usually not symmetric in nature.
Astrocytoma of the spine is an intramedullary glioma, more often located in the cervical than thoracic spine. This lesion presents as a hyperintensity on proton density and T2 weighted imaging, and almost always enhances. On T1 imaging, there is cord expansion, usually less than four segments. Again this is distinguished from SCD by location (intramedullary T2 signal intensity versus dorsal and lateral columns) and contiguity (SCD is contiguous over multiple vertebral segments).
Spinal cord infarction causes permanent tissue loss in the spinal cord secondary to vessel occlusion and usually presents hyper acutely. On MRI imaging, focal T2 hyperintensities can be seen in the gray matter, gray matter with adjacent white matter, or an entire cross section of the cord. These lesions are usually within the thoracic cord as it is an arterial border zone. T2 hyperintensities may also bee visualized within the anterior vertebral body bone marrow or deep medullary portion near the endplate secondary to vertebral body infarction. Diffusion weighted imaging shows restricted diffusion in the affected areas of the cord. Slight cord expansion and decreased signal is noted on T1 imaging. The MRI lesion distribution, bony involvement and diffusion changes differentiate spinal cord infarction from SCD.
Infectious myelitis can be secondary to HIV vacuolar myelopathy, varicella-zoster/herpes, or Lyme disease. On T1 imaging, there is cord expansion that nearly fills the spinal canal and variable, nonfocal enhancement status post contract administration. On T2 imaging, there is diffuse increased signal intensity throughout the involved segment, secondary to a swollen edematous cord. Unlike SCD, the signal abnormalities are not limited to the spinal tracts. However, MRI imaging findings may be identical to B12 deficiency and in such cases, can be distinguished by clinical presentation and laboratory findings.