Findings
FindingsOn CT imaging, there is a well circumscribed predominantly cystic lesion at the level of the hyoid within the right neck soft tissue. The lesion has a sharp margin with the sternocleidomastoid muscle and is located superficial to the carotid artery. No adjacent stranding or inflammatory changes.
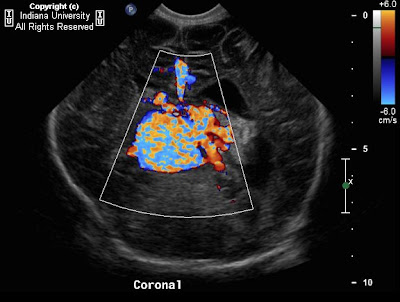
On ultrasound imaging, an anechoic structure with small amount of debris is noted within the right neck with some internal septations. No flow is noted within the lesion.
Differential diagnosis:
- Branchial cleft cyst
- Lymphangioma
- Thymic cyst
- Suppurative jugulodigastric node
- Cystic vagal schwannoma
- Cystic malignant adenopathy
Differential diagnosis considerationsLymphangioma are multilocular, trans-spatial collections that fill the available spaces. If they are unilocular and located in typical position of second branchial cleft cyst, it may be very difficult to differentiate between the two. One characteristic that helps differentiate the two, is that lymphangiomas are infiltrative in nature while branchial cleft cysts are well defined and round.
Thymic cysts are usually located inferior within the cervical neck and are centered in the lateral visceral space.
Suppurative jugulodigastric node presents as a thick walled ovoid mass at the angle of the mandible.
Cystic vagal schwannoma has a thick, enhancing wall and is usually centered behind the carotid space causing displacement of the internal jugular vein and internal carotid artery.
Cystic malignant adenopathy presents as a necrotic mass with a thick, enhancing wall. Cystic metastasis (e.g. papillary thyroid carcinoma) and necrotic metastases (e.g. squamous cell carcinoma) as well as necrotic tuberculous lymphadenitis can have a cystic appearance similar to second branchial cleft cyst, therefore it is very important to differentiate the two and ultrasound fine needle aspiration with fluid analysis can provide useful information.
Diagnosis: Branchial cleft cystDiscussionThe branchial apparatus is a precursor to many head and heck structures. The second branchial cleft arch overgrows the second, third, and fourth clefts and forms a cavity called the cervical sinus. During the seventh week of gestation, the cervical sinus is normally obliterated. However if there is incomplete involution, a congenital squamous epithelial lined cyst (branchial cleft cyst), sinus tract or fistulae can form. There are four subtypes of branchial cleft cyst, described below, of which greater than 90% are second branchial cleft cyst.
Classification
- Type I: anterior to the sternocleidomastoid, beneath the platysma muscle.
- Type II: adjacent to the internal carotid artery, often adherent to the internal jugular vein.
- Type III: extends between the internal and external carotid artery to the lateral pharyngeal wall.
- Type IV: lies against the lateral pharyngeal wall and may extend into the skull base.
Majority present as a painless, compressible lateral neck mass; however, they can become tender, enlarged, inflamed or develop abscesses, especially during periods of upper respiratory tract infection.
The size of cysts are variable and range from a few centimeters to > 5 cm. Most are ovoid or rounded in shape; some present with a focal rim of cyst extending to the carotid bifurcation, which presents as the "Notch sign" which is pathognomonic for second branchial cleft cyst.
These cyst can occur anywhere along the line from the tonsillar fossa to the supraclavicular region and more commonly are seen in the following areas:
- Posterolateral to submandibular gland, most are at or immediately caudal to the mandible angle.
- Lateral to carotid space.
- Anteromedial to sternocleidomastoid muscle.
- Some unusual locations for cyst include:
- Superiorly within parapharyngeal space or carotid space.
- Inferior along anterior surface of infrahyoid carotid space.
- Fistulous tract between the external and internal carotid arteries to faucial tonsil.
Treatment is usually surgical resection.
Controversy exists as to the possibility of carcinoma arising within the branchial remnant squamous epithelial lining or not. However, metastatic squamous cell carcinoma to regional lymph nodes that masquerades as branchial cleft cyst is more common.
Imaging findingsCT or MRI an easily suggest diagnosis. Contrast is used to help differentiate cystic from solid mass.
Contrast enhanced CT:
- Low density cyst with nonenhancing wall and surround soft tissue if noninfected.
- If infected, wall appears thicker and enhances with surround soft tissues appearing as dirty cellulites.
MRI:
- On T1W imaging: cyst is isointense to CSF. If cyst is recurrently infected, it may contain hyper intense contents secondary to increase protein concentration.
- T2W imaging: Hyperintense cyst, no discernible wall.
- FLAIR imaging: Cyst is iso or hyper intense to CSF.
- T1W imaging + Contrast: Peripheral wall enhancement if infected.
Ultrasound:
- Anechoic thin-walled cyst with through transmission.
- May be hyper echoic with internal debris or even pseudo-solid in appearance.
- If infected, see thickened cyst wall.