


Findings:
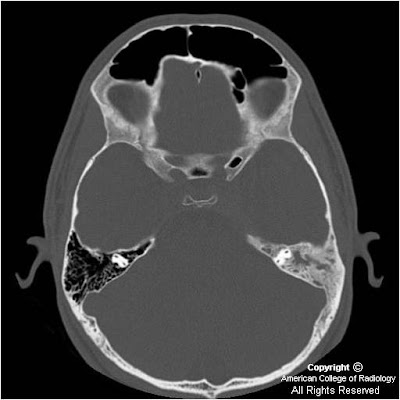
Figure 1and Figure 2: There is diffuse sclerosis of the left mastoid with near complete opacification of the mastoid air cells.
Figure 3 and Figure 4: There is diffuse sclerosis of the mastoid with a soft tissue thickening identified in the cavernous sinus. There is additional dural extension suggested along the left leaf of the tentorium.
Diagnosis: Langerhans cell histiocytosis (LCH), skull base
Langerhans cell histiocytosis (LCH) is the term used to describe the spectrum of diseases characterized by proliferation of specialized bone marrow-derived dendritic cells, called Langerhans cells (LC) with bone and/or soft tissue involvement. Although the etiology and pathogenesis of LCH is poorly understood, studies suggest that it is a reactive disease caused by immune dysregulation. The disorders once collectively called Histiocytosis X (eosinophilic granuloma, Hand-Schüller-Christian disease, and Letterer-Siwe disease) are now part of this category. LCH is defined in terms of whether it involves a single site, multiple sites, a single organ, or multiple organs. Prognosis and treatment are closely linked to the extent of disease at presentation and whether or not “risk” organs (ie: liver, spleen, lung, bone marrow) are involved. In general, patients with LCH localized to the bone have a favorable prognosis whereas patients with multisystem involvement incur a poor prognosis.
Langerhans cell histiocytosis of the bone is a relatively rare disorder with an incidence of one case per 2 million persons per year. The skull is the most commonly involved bony site in the pediatric age group. Otologic involvement usually occurs during the course of multisystemic disease, and may be the only symptom in up to 25% of patients. Because the otologic symptoms are similar to otomastoiditis, diagnosis of LCH of the skull is often delayed. The most frequent otologic symptom is otorrhea resistant to medical treatment. Other findings include mastoid swelling, aural polyps, periauricular eczema, sagging of the canal wall skin from erosion of the posterior bony external auditory canal, conductive hearing loss, and otalgia. Neurosensorial deafness, vertigo from involvement of bony labyrinth, and paralysis of cranial nerves (particularly VII and VIII) have been described. The classic clinical profile is a young man with otalgia, otorrhea, and post-auricular mass. If the diagnosis is suspected, a full examination and work-up is warranted to exclude the possibility of multisystemic or multiostotic disease.
The best imaging modality to diagnose LCH of the skull base is dedicated temporal bone CT. The skull base may show focal or diffuse bone destructive change and may present as: 1) sharply-defined “punched out” appearance, 2) irregular sclerotic lesions or 3) fragments of bone within soft tissue component. Other locations that are affected include the petrous apex, mandible, maxilla, and vertebral body. Ossicular and otic capsule destruction are common. Bilateral disease occurs in up to 30% of patients. The radiologic differential diagnosis for LCH of the skull base includes mastoiditis, cholesteatoma, cholesterol granuloma, and rhabdomyosarcoma. Rhabdomyosarcoma may exactly mimic unilateral LCH- biopsy may be required to differentiate the two entities.
Contrast-enhanced examination may help differentiate inflammatory mastoid lesions from LCH. Findings include a heterogeneously enhancing mass in the temporal bone, including mastoid complex, middle ear, extracalvarial, intracranial, and extra-dural compartments. The skull base tends to show a variable amount of enhancing soft tissue. On MR, T1 weighted images reveal iso- to hypointense mastoid, middle ear or skull base mass. T2 weighted images reveal hyper- to isointense soft tissue mass. Contrast-enhanced T1 weighted images show a heterogeneously-enhancing mastoid, middle ear, or skull base mass. MR may be obtained to delineate boundaries of soft tissue component which generally displays poorly defined borders.
The prognosis of LCH localized to the temporal bone is excellent, with a cure rate of 90%. The soft tissue component resolves initially, followed by reossification of the bone lesion. The treatment of choice for LCH localized to the mastoid-middle ear consists of surgical curettage or mastoidectomy. Low dose radiotherapy is used for larger, destructive lesions. Systemic disease is treated with chemotherapy in combination with radiation therapy.