





Findings
Axial non-enhanced CT image demonstrates ill-defined slightly hyperdense lesion (Figure 1) centrally within the posterior fossa.
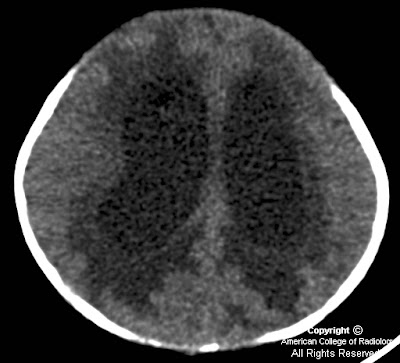
Axial non-enhanced CT image shows hydrocephalus with transependymal flow of CSF (Figure 2).
Axial T1-weighted image (Figure 3) demonstrates a posterior fossa mass that is hypointense on T1.
Axial T2-weighted image shows a well-defined heterogeneous mass (Figure 4).
Axial ADC map image (Figure 5) shows that the solid portions of the mass are dark, consistent with relatively decreased diffusion of water molecules.
Axial T1-weighted postcontrast image (Figure 6) shows a mass with heterogeneous contrast enhancement and areas of probable cystic change.
Sagittal T1-weighted postcontrast image demonstrates the large mass causing herniation of cerebellum and cerebellar tonsils inferiorly through the foramen magnum (Figure 7). The brainstem is compressed and shifted ventrally. There is compression of cerebral aqueduct that leads to marked hydrocephalus.
Differential diagnosis:
- Medulloblastoma
- Juvenile pilocytic astrocytoma
- Ependymoma
- Atypical teratoid
- Rhabdoid tumor
Diagnosis: Medulloblastoma
Medulloblastoma is thought to arise from undifferentiated neuroepithelial cells by neoplastic transformation of cells in the roof of the fourth ventricle and is categorized as a primitive neuroectodermal tumor (PNET). Medulloblastomas are the most common malignant central nervous system in children and the second most common pediatric brain neoplasm. It is one of the two most common primary tumors of the posterior fossa in children, the other one being juvenile pylocytic astrocytoma (JPA). Medulloblastoma is almost always found in the cerebellum, typically arising from the vermis. The tumor most frequently occurs in males under 10 years of age. Common presenting symptoms are headache, vomiting, and ataxia, and they are usually of less than 3 months in duration.
The classic CT appearance of medulloblastomas is a hyperdense, well-circumscribed, homogeneously-enhancing central cerebellar mass associated with obstructive hydrocephalus. On MR imaging, the tumors are typically of hypointense T1 signal and isointense to hypointense on T2-weighted images. There is a greater degree of heterogeneity among medulloblastomas on MR images compared to CT scans. Nearly all tumors show heterogeneous enhancement following gadolinium. The tumor appears heterogeneous due to hemorrhage, cystic change, and calcification, which occurs in 20% of cases. Medulloblastomas consist of small, densely packed cells (one of the "small blue cells" tumors), which leads to relatively decreased diffusion of water molecules seen as hypointensity on ADC maps. This feature appears to distinguish these tumors from JPA, which contain large interstitial spaces leading to increased diffusion and hyperintensity on ADC maps. Taurine peak may be detected within medulloblastomas on MR proton spectroscopy. Evidence of subarachnoid metastatic spread is present in up to one-third of cases at initial diagnosis.
Leptomeningeal involvement of the spinal cord is the most common site of spread and contrast-enhanced MRI is the imaging study of choice. Characteristic findings of spinal cord involvement are nodular enhancement of the cord surface or nerve roots, clumped nerve roots, and diffuse enhancement of the thecal sac. The use of ventriculoperitoneal shunts may lead to metastatic spread in the abdominal cavity. Tumor is very radiosensitive and thus, a combination of surgery and radiation treatment is most commonly used. Imaging of the entire brain and spinal cord is important to guide treatment prior to surgery. In addition to surgery and radiation, chemotherapy and shunt placement are also used in some cases, and postoperative chemotherapy without radiation has recently been found promising. A follow-up MRI with contrast is obtained postoperatively within 48 hours to assess for residual tumor before the development of enhancing reactive gliosis, which could be confused as tumor. Postoperative surveillance imaging of the brain and spine is regularly preformed, as recurrence is frequent. The majority of recurrences occur in the first two years after treatment. Recurrence presents as leptomeningeal enhancement or focal parenchymal nodular enhancement within the brain, most frequently in the posterior fossa, and metastatic involvement of vertebral bodies is sometimes seen.
Nessun commento:
Posta un commento